
News
Behind the Headlines
Two-Cents Worth
Video of the Week
News Blurbs
Articles
Testimony
Bible Questions
Internet Articles (2015)
Internet Articles (2014)
Internet
Articles (2013)
Internet Articles (2012)
Internet Articles (2011)
Internet Articles (2010)
Internet Articles
(2009)
Internet Articles (2008)
Internet Articles (2007)
Internet Articles (2006)
Internet Articles (2005)
Internet Articles (2004)
Internet Articles (2003)
Internet Articles (2002)
Internet Articles (2001)


 n
April 29, 2008 the widow of a man who died after receiving contaminated
Heparin in a dialysis treatment was among the family members who testified
before the US House Subcommittee on Oversight and Investigations. In the
same hearing, FDA officials also testified that contaminated Heparin was
responsible for 81 deaths in the United States and, in the view of the
drug regulator, Dr. Janet Woodstock, director of the agency's drug
center, the contamination was deliberate. Woodstock said: "The
FDA's working hypothesis is that this was intentional contamination, but
this is not yet proven." Sadly, after several more deaths, it
was. One of the US companies selling Heparin to hospitals, doctors and
medical treatment facilities, Robert Parkinson, head of Baxter
International, also testified. Parkinson agreed with Woodstock's
assessment and said his company was "...alarmed that one of [their]
products was used in what appears to have been a deliberate scheme to
adulterate a lifesaving medication."
n
April 29, 2008 the widow of a man who died after receiving contaminated
Heparin in a dialysis treatment was among the family members who testified
before the US House Subcommittee on Oversight and Investigations. In the
same hearing, FDA officials also testified that contaminated Heparin was
responsible for 81 deaths in the United States and, in the view of the
drug regulator, Dr. Janet Woodstock, director of the agency's drug
center, the contamination was deliberate. Woodstock said: "The
FDA's working hypothesis is that this was intentional contamination, but
this is not yet proven." Sadly, after several more deaths, it
was. One of the US companies selling Heparin to hospitals, doctors and
medical treatment facilities, Robert Parkinson, head of Baxter
International, also testified. Parkinson agreed with Woodstock's
assessment and said his company was "...alarmed that one of [their]
products was used in what appears to have been a deliberate scheme to
adulterate a lifesaving medication."
Family members
gave the attentive Congressmen heartwrenching stories of the last moments
in the lives of their loved ones. Johanna Marie Staples of Toledo,
Ohio said her husband, Dennis, was looking forward to celebrating
his 60th birthday at a family gathering when he completed his dialysis
treatment that day. What was supposed to be a celebration of life became the mourning of Dennis'
death.
What was supposed to be a celebration of life became the mourning of Dennis'
death.
Colleen Hubley, a dialysis nurse reported how her husband, Randy, came home from his dialysis treatment experiencing diarrhea, abdominal pain and he was having trouble breathing. He died the following evening after she tried to save his life with cardiopulmonary resuscitation. Leroy Hunley, also of Toledo, described to Congress how he lost both his 65-year old wife and his 47-year old son within weeks of each other. Both suffered from genetic kidney disease that required regularly scheduled dialysis treatments. During those treatments, Heparin was routinely used.
The Food & Drug Administration [FDA] is once again in conflict with its own Congressional mandate to protect the American people from unscrupulous merchant princes who line the pockets of politicians with gold. China has proven over and over again that it will not abide by international food product or pharmaceutical safety rules. It should be abundantly clear to the American people by this time that saving a few cents to a few dollars per product to buy goods made in the 3rd world—particularly in Communist China (which is using your international trade dollars to upgrade their weapons systems sufficiently to destroy us)—may put their life at risk.
In July, 2007 over a hundred pet owners in the United States lost both canine and feline pets who had ingested high levels of a thermoset plastic, melamine, and also toxic levels of cyanuric acid that had been added to inferior grades of both rice and wheat gluten to make the gluten appear that it contained high levels of healthy protein when it had almost none. Melamine, when combined with formaldehyde, becomes a synthetic polymer resin used to make things like Melmac™ dishes and Formica™ counter tops. Melamine is not edible.
Why would the Chinese put melamine into the wheat and rice gluten they were selling to the industrialized nations? Because US, European Union and Canadian food products buyers do not chemically test the gluten samples they inspect to determine the protein levels. They judge the protein levels of the samples by their appearance only. For the Chinese, who are accustomed to high profits from using slave labor, using inferior grades of rice and wheat gluten simply increased their profit margins. (And you never thought that communists could love the free enterprise system.)
Over 50% of the trade goods rejected for entry into the United States by the Consumers Product Safety Commission [CPSC] are imports that were made, or processed, in China. In the first quarter of 2007, FDA and USDA inspectors rejected 298 food product shipments from China. Among them were baby toys painted with lead-based paints, children's jewelry made from lead (lead poisoning is one of the causes of autism). One victim was a 4-year old boy who swallowed the charm from a Chinese-made charm bracelet. The charm was 99% lead layered with silver. The toddler died. Nancy Nord, head of the CPSC said that from 2000 to 2005 over 20 thousand children have been treated at local hospitals for lead poisoning. Most of them swallowed jewelry elements.
Even though China persists in poking America in the eye with a sharp stick, this nation's greedy merchant princes ignore the danger that China's products increasingly pose for US consumers, and to make an extra buck or two on the products they sell, are buying even more unsafe products from a nation which remains our enemy. The latest assault on the unsuspecting consumers in the United States came in the form of a contaminated blood-thinner, Heparin™, used to treat heart attack and stroke patients.
In January, 2008, reports of 350 adverse allergic-reaction events resulting in 19 deaths from contaminated Heparin™ reached the FDA. The alert came from Baxter, International, a major supplier of Heparin™ in the United States. The raw Heparin™ component sold to Baxter International was manufactured by a large, unregulated Chinese manufacturer, Changzhou SPL Co. Ltd., of Changzhou City, Jiangsu, China. Changzhou SPL partners with Scientific Protein Laboratories, LLC of Waunakee, Wisconsin through a Joint Venture. SPL, formerly Tyco Heathcare, is a leading global supplier of active pharmaceutical ingredients [APIs] that are utilized by pharmaceutical companies to produce injectable solutions to medical problems. Changzhou SPL is not regulated by the Chinese government because, while it is partially-owned by the Chinese government, it is also partially-owned by SPL in the United States.
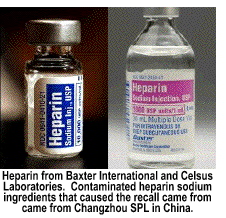 And,
because Changzhou SPL is tied to an American company, the
FDA did not feel it was necessary to inspect its plant in Changzhou—a
violation of FDA protocol. Because bureaucrats bowed to political pressure
from Congressmen who bowed to pressure from the lobbyists of the pharmaceutical
industry, 19 people died in the first disbursement of tainted Heparin™.
SPL heparin sodium USP, an odorless white amorphous hygroscopic powder,
is the active ingredient used to produce serum Heparin™. It is sold
to companies like Baxter International, Celsus Laboratories
Inc. of Cincinnati, Atrium Medical Corporation and Medtronic,
Inc. who also bought SPL heparin sodium from Changzhou SPL.
And,
because Changzhou SPL is tied to an American company, the
FDA did not feel it was necessary to inspect its plant in Changzhou—a
violation of FDA protocol. Because bureaucrats bowed to political pressure
from Congressmen who bowed to pressure from the lobbyists of the pharmaceutical
industry, 19 people died in the first disbursement of tainted Heparin™.
SPL heparin sodium USP, an odorless white amorphous hygroscopic powder,
is the active ingredient used to produce serum Heparin™. It is sold
to companies like Baxter International, Celsus Laboratories
Inc. of Cincinnati, Atrium Medical Corporation and Medtronic,
Inc. who also bought SPL heparin sodium from Changzhou SPL.
On February 12, 2008 as both the death count and number of "allergic reaction" events increased, the FDA released an advisory to physicians and hospitals to stop using Baxter's Multiple-dose Heparin™ and to use other suppliers until they discovered what was causing allergic reactions and dangerously low blood pressure in hundreds of patients. Baxter International stopped manufacturing Heparin™, and on Feb. 28 the FDA notified doctors and hospitals that Baxter had voluntarily recalled their multi-dose and single-dose vials of heparin sodium for injection as well as their heparin lock flush solutions.
By April, over 1,000 people had the symptoms. The death toll reached 81 people. It would not be until May 15, 2008 that Atrium and Medtronic (who do not produce injection Heparin™) would initiate voluntary, and what they called "precautionary," recalls of some of the products they do create. Atrium recalled selected lots of Hydraglide™ brand heparin-coated thoracic drainage catheters. Medtronic withdrew selected products used during cardiopulmonary bypass heart surgery. In addition, Medtronic advised doctors and hospitals that selected lots of Trillium-coated products were manufactured with heparin coatings. (The FDA noted that, based on current data, the benefits of using the affected products outweigh any potential risk to patients.)
A month earlier—in March—
the FDA figured out that the Changzhou SPL heparin sodium,
a highly sulfated glycosaminoglycan, was being adulterated with a heparin-like
chemical called over-sulfated chondroitin sulfate, a white powder
harvested from shellfish. It appears the over-sulfated chondroitin
sulfate was added as a filler to cut the cost of heparin sodium in
powder form.  Apparently
the chemists at the Chinese facility, knowing that chondroitin sulfate
was also a glycosaminoglycan, thought it would be a harmless additive
that would substantially cut the cost of the drug they were selling to
American pharmaceutical companies without causing an adverse affects.
Apparently
the chemists at the Chinese facility, knowing that chondroitin sulfate
was also a glycosaminoglycan, thought it would be a harmless additive
that would substantially cut the cost of the drug they were selling to
American pharmaceutical companies without causing an adverse affects.
The presence of this synthetic chemical suggested to the FDA that the heparin sodium manufactured at Changzhou SPL might be tampered with deliberately, but at the time the FDA could not determine at what point in the manufacturing process the chemical was added to the heparin sodium compound. The Bush-43 FDA suspected the contamination occurred at Changzhou SPL but refused to tell Congress the names of any company in China in may have supplied the adulterated heparin ingredients to the United States. The office of Dr. Andrew von Eschenbach, MD, Commissioner of the FDA through his spokeswoman, Kathleen Quinn told the media that confidentiality agreements with vaccine and serum producers bar the FDA from revealing their names.
According to 8th term Congressman Bart Stupak [D-MI] Congress has been at odds with the FDA from the beginning over the manner they chose to deal with this problem. "The FDA thinks they have this under control," Stupak said, "but they really don't. If I was the FDA Director, I'd shut down every drug coming in from China until each one had individually proven to be safe."
The FDA countered that it does not have the legal authority to prevent the importation of Chinese-manufactured drugs, citing a Memorandum of Agreement that regulates relations between the FDA and China. The Memorandum was created to placate the pharmaceutical industry by speeding up the process—and, conversely, sacrificing the thoroughness—of FDA inspections of Chinese drug facilities. Stupak countered by criticizing the Memorandum which, he said, is "useless." Stupak noted that the agreement does not give the FDA any real access to Chinese factories with what amounted to as "walk-throughs." The FDA admitted that it had inspected a number of Chinese heparin ingredient factories in February. Changzhou SPL was one of the plants that was visited. The FDA said it was not given any access to workers, records or workshops. An FDA compliance official told Congress that another inspection could be attempted, but "...I cannot say whether they will admit us or not, or whether they will allow us to do a full inspection." In reality, Stupak said, the FDA teams can go to China and scrap paint off the walls to inspect all they want. They just can't inspect the labs where the heparin ingredients are processed.
 China
has objected to allegations that their country is the source of the contamination
pointing out, in early March, that the drug could just as easily have
been tampered with at Baxter's Cherry Hill, New Jersey plant.
The Chinese Food and Drug Administration accused Baxter International
of failing to cooperate with their investigation into the contamination.
Chinese health officials said Baxter International, and
its CEO Robert L. Parkinson, Jr., refused to surrender samples
of SPL heparin sodium and the heparin ingredients that Baxter
purchased from other sources to them in April so they could test all of
the samples from all sources for contamination.
China
has objected to allegations that their country is the source of the contamination
pointing out, in early March, that the drug could just as easily have
been tampered with at Baxter's Cherry Hill, New Jersey plant.
The Chinese Food and Drug Administration accused Baxter International
of failing to cooperate with their investigation into the contamination.
Chinese health officials said Baxter International, and
its CEO Robert L. Parkinson, Jr., refused to surrender samples
of SPL heparin sodium and the heparin ingredients that Baxter
purchased from other sources to them in April so they could test all of
the samples from all sources for contamination.
Powerless to regulate American partnerships in China, the FDA has had to rely on US companies voluntarily testing the substandard pharmaceutical ingredients they purchase from China. Yet that testing failed to detect the heparin contamination at Baxter's facility in January and February, 2008. "It was indistinguishable from the real heparin," Parkinson told Congress. However, in the aftermath of the heparin sodium problem, Parkinson said, Baxter International has developed a new testing method that can distinguish between pure heparin and oversulfated chondroitin sulfate. How? They test for oversulfated chondroitin sulfate and not heparin. What that means is that as soon as the Chinese chemists find another cheap heparin look-alike (which they assume should work the same way for less money), they will try it. Communist China, which is new to the free enterprise system, thinks that anything that fools the consumers is doable—even if a few consumers die in the process. After all, China is a country with far too many consumers. So a few hundred less can only be construed as an unexpected caveat. However, where the Chinese may be a little cavalier about reducing the population with contaminated products, that practice doesn't bode well in the United States.
And therein lies the problem. American industrialists, eager to escape the most rigid safety guidelines in the world in the United States used NAFTA to escape the ever-watchful eye of Big Brother by partnering with companies in communist China. In the case of Changzhou SPL, Scientific Protein Laboratories, LLC (formerly Tyco Heathcare), is the majority stakeholder in that partnership. Yet, the FDA can't honestly inspect that factory since the minority shareholder in the Chinese government (or members of that government who are high enough in the Party that Changzhou SPL is immune from harassment from the mosquitoes in the United States government.)
One might think because the FDA was on the problem in February (the first contaminated heparin ingredients arrived at Baxter's Cherry Hill, New Jersey plant in November, 2007), and because Baxter, Atrium and Medtronic initiated voluntary recalls that the problem is over. But that's simply not the case. On November 6, 2008 the FDA seized contaminated batches of Heparin™ from Cincinnati-based Celsus Laboratories. Seized were 11 lots of tainted blood thinner. Celsus, like Baxter, learned that the heparin ingredients they received from China were contaminated. Celsus informed its customers that Heparin™ shipped from their facility between January and April, 2008 could be contaminated. However, according to FDA spokeswoman Karen Riley, Celsus did not tell their customers to destroy the product or send them back. The tainted Heparin™, she said, was sold to universities and research-supply facilities in Australia, Canada, Europe, Japan, and the United States. "This was a contaminated product," she said,"and we don't think that they did enough. We decided to seize the remainder and destroy it."
The FDA noted that there are no reports of anyone taking the Celsus heparin injectable product dying or suffering from allergic reactions. But then, if any hospitals and medical treatment facilities who injected a heart attack or stroke patient, or those on dialysis, or anyone needing Heparin during an operation with contaminated Heparin™ without knowing the product was tainted, they would naturally assume the patients who died—if any—were victims of their ailments and not of the medication that should have saved their lives.
It is critical that US consumers look at the country of origin before buying anything. If that product is made in China, be wary. China is notorious for taking shortcuts in product creation. Other than the Heparin™ controversy, its important to remember the last two scares concern human foods laced with melamine to make the products appear healthy when the opposite was true.
First, and most recent was the Canadian chocolate-melamine scare. Yes, the same thing that killed your pets last year targeted Canadian children this year. Yet another unidentified Chinese factory delivered a shipment of 3,360 kilograms of chocolate coin candy under the brand name Sherwood Brands Pirate's Gold Milk Chocolate Coins that were sold to Costco (in Canada only) and several dollar stores across Canada. The affected candy has bar codes of 0-36077, 112407 and lot code 192851. The melamine-laced chocolate was sold to Canadian retailers between January and April, 2008.
In September the FDA warned US consumers not to feed any baby formulas made in China to infants. Again, the baby formula was linked to melamine contamination as well. Infant formula made in the United States is safe, but you would be advised to check any containers you purchase to make sure they are not made in China. Melamine tainted baby formulas is a serious problem in China. To date over 54 thousand Chinese babies have fallen ill due to malamine-laced baby formula. Four babies have died.
It's time that US consumers wake up to the problems caused not only by inferior consumer products that are made in China, but of dangerous consumable products made there as well. Saving a dollar in hard times is not worth the risk of losing the life of a loved one at anytime. If you can't afford to pay a few dollars more for a product made in this country, do without it. If enough people leave the Chinese goods on the store shelves, America's merchant princes will eventually get the message. When they start shopping for the products they need to stock their shelves in the United States instead of the third world, US jobs will increase, the economy will improve and we will have guaranteed that our children and our children's children will have a chance to share the great American dream before the movie comes out—in Chinese.

Copyright © 2009 Jon Christian Ryter.
All rights reserved.


